Spontaneous Coal Combustion Part 1: A Burning Mystery

This blog post is part one of a three-part series on spontaneous coal combustion. In this post, we focus on the history and research, and in the upcoming posts we’ll take a look at preventative measures and best practices.
When coal burns, it releases carbon dioxide, water, heat, sulfur, particulate matter, and other compounds into the air, while leaving behind ash that is laden with silica, mercury, cadmium, arsenic, and other compounds. When this combustion occurs inside a controlled environment under controlled conditions, such as in a coal-fired power plant, the emissions can be scrubbed of toxins and the ash can be contained and properly managed. However, when this combustion occurs in uncontrolled environments (i.e. coal mines, piles, silos, barges, seams, etc.), these toxins are freely released into the environment. While the direct casualties and damages of these fires are relatively small, the indirect and cumulative effects of this uncontrolled burning of coal are monumental. So, what causes this uncontrolled burning?
There are several reasons coal may burn in uncontrolled environments:
- Lightning strikes: A January 2006 West Virginia coal mine explosion that claimed 12 lives was the deadliest U.S. fire started by lightning in recent years.
- Carelessness or accidents: A trash fire in a landfill in Centralia, Pennsylvania ignited a vein of anthracite in 1962, and that fire is still burning to this day.
- Spontaneous combustion: Coal has a tendency to self-heat due to the auto-oxidation of its compounds, making spontaneous combustion the most common cause of uncontrolled burning. There are thousands of uncontrolled coal fires across the earth, some of which have been burning for thousands of years.
What is the geographical occurrence and history of spontaneous coal combustion?
Because spontaneous combustion is the most significant cause of coal fires in uncontrolled environments, it has been the focus of a lot of research and development, especially in the last 10-20 years. It is a problem that doesn’t just plague the US coal industry, it is a global issue that affects the health and well-being of everyone on Earth, regardless of proximity to coal.
In Australia, the oldest known coal fire has been burning for 6,000 years near Mount Wingen (also known as Burning Mountain).

Burning Mountain was part of a vast territory owned by the Wanaruah people, and now showcases slowly combusting coal seams about 30m below the ground. Source: Australia National Parks and Wildlife Service
In China, it is estimated that up to 200 million tons of coal burn each year in uncontrolled environments due to spontaneous combustion. That equates to as much as 3% of the total global greenhouse gas emissions.

An area of more than 310,000 square meters is burning at the Urumqi mine. The fire causes an estimated 217,000 tons of coal to be wasted annually and makes some 11.28 million tons of coal inaccessible for mining.
India has the largest concentration of coal fires, and it is estimated that 80% of those fires started due to spontaneous combustion. Indian coal fires were first documented in coal fields located in Raniganj back in 1865. In addition, mines in Jharia, Jharkhand are famous for their rich coal resources, but records show nearly 70 fires have been burning for over 100 years dating back to 1916. Coal fires have caused the Indian government to spend nearly $1 billion to relocate villages away from the mines in this region. It is estimated that these fires have burned more than 37 million tons of coal. While India is having success in controlling many fires, the blazes that rage on are obstructing nearly 1.4 billion more tons of coal from being safely mined.

Coal fires burn early in the morning in January 2014 at a privately owned coal mine in India’s Jharkand state, making the air dense with noxious smoke and dust. Forty percent of India’s mineral wealth lies in Jharkhand. Source: NPR.org
In Indonesia, enormous peat (a low energy form of coal) fires flare up each year sending so much smoke into the air that it affects visibility and health in neighboring countries.

Heavy smoke pours from peat fires in Indonesia, blanketing six countries. Picture: NASA
There are thousands of other well-documented coal fires in dozens of other countries, and most of them are attributed to spontaneous combustion. Even in the United States of America, home to some of the largest coal reserves in the world, there are nearly 200 reported active coal fires, and likely many more which go unreported. Some underground coal seam fires in the Powder River Basin region of Wyoming and Montana can even be dated back over 4 million years!
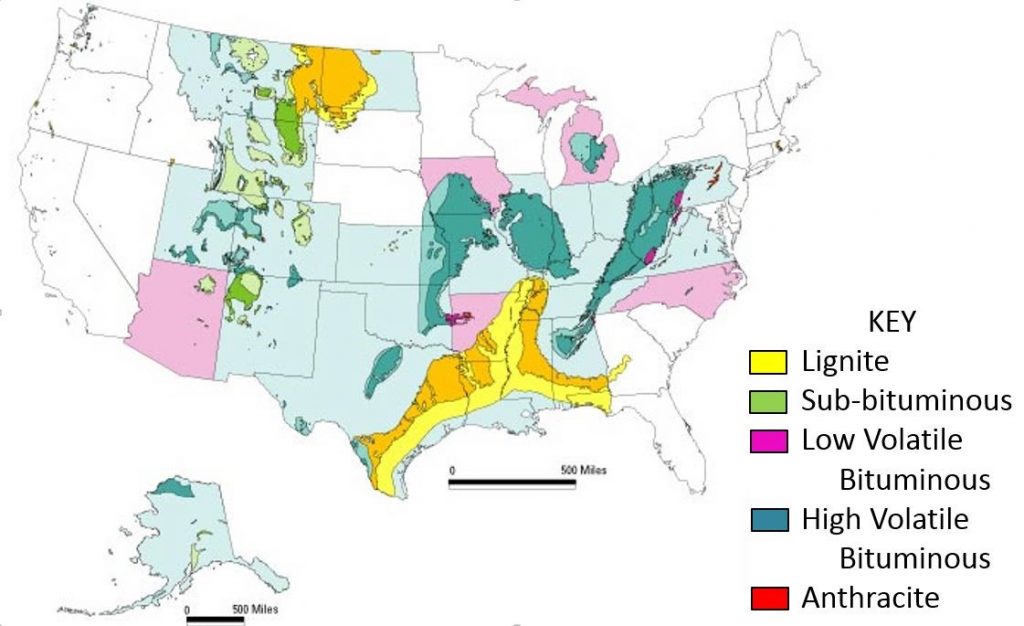
A map of the coal reserves located in the United States by region and type
Source: US Geological Survey (USGS). Coal: A complex natural resource
Seen across the northern Great Plains of the US, naturally burning coal-bed fires over time have baked and fused colorful, reddish sediments of clay, shale, and sandstone rocks into clinker, a form of natural brick.

Clinker pit south of Rhame, North Dakota. Source: North Dakota Geological Survey
These clinker beds range from 40 feet in North Dakota to much thicker in the PRB region of Wyoming and Montana. Over the years, these clinker seams have resisted erosion, thereby forming much of the landscape in the western US.
Early western explorers, Lewis and Clark, even wrote about these formations in their journals during the spring of 1805. They recorded evidence of fire, coal seams, rich hard clay, and where these seams meet together, burnt appearances. Clark originally named one river the Redstone River, due to the rich reddish seams of clinker, but it was later renamed to Powder River because the smell of coal fires reminded settlers of the smell of burning gunpowder. As settlers and explorers continued their move west, reports of coal fires in the northern Great Plains only grew, many of which have been caused by spontaneous coal combustion.

Smoldering coal pile, found in the coal yard at a coal-fired power plant burning PRB coal, being reworked
In the last 150 years, human mining activities and erosion have intensified these fires, exposing thousands of acres of fresh coal to the air. As mentioned earlier, when coal is exposed to oxygen, the result is auto-oxidation, and the resulting chemical reaction produces heat. When this happens in uncontrolled environments, the process can occur slowly over many years or quickly in just a few hours, depending on the conditions. As the heat is released, it intensifies the reaction, producing more heat, and initiating a perpetual cycle. Once there is enough heat, spontaneous combustion occurs. When this happens underground in coal seams, these fires form large fissures, and as long as the heat is able to generate faster than it can dissipate, this spontaneous combustion can burn and smolder for decades without showing signs on the surface.
What is spontaneous combustion?
To be able to prevent or control spontaneous combustion, it is important to understand exactly what is happening at a chemical and physical level. To begin, spontaneous combustion is a naturally occurring phenomenon caused by coal oxidation, a chemical process which produces heat, even at ambient air temperature. This process is a natural balance of heat formation and dissipation and if this heat is allowed to accumulate, the oxidation rate increases exponentially, creating more and more heat until the point of thermal runaway (point of no return), resulting in a spontaneous fire.
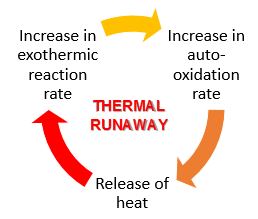
The exact mechanism of spontaneous coal combustion process is still being debated, but researchers have been investigating it since 1908 and most researchers agree that oxidation plays a major role in this process. While the complex chemical nature of coal is not fully understood, the general chemical/physical reaction is as follows:
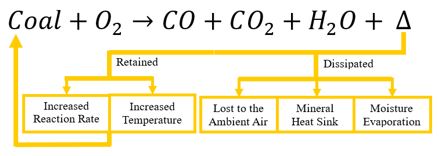
Heat Flow During Coal Auto-Oxidation
When sufficient oxygen is available at low, ambient temperatures, the reaction with coal is exothermic and produces heat. The heat, shown as Δ in the above equation, can be either dissipated or retained. In order for the coal body temperature to remain static, the heat from this reaction must be dissipated to surrounding substrates (i.e. soil, minerals, air, etc.) like a heat sink, or the heat must be transferred via moisture loss or evaporation. It is when this heat cannot be dissipated that the coal body temperature begins to rise, increasing the rate of the reaction exponentially and inevitably causing spontaneous combustion.
Studies that estimate the rate of this phenomenon show that for every 18°F rise in temperature the reaction rate can double. That means if ambient coal temperatures were to rise from 86°F to 122°F, the rate of this reaction would quadruple (4X). If allowed to rise further to 158°F, this reaction would happen 16X faster!
While it is unlikely that we’ll ever be able to prevent spontaneous combustion that occurs naturally in underground coal seams, we do understand enough about the process to prevent spontaneous combustion during the mining, transferring, and storage of coal. What this ultimately means for coal mines, coal trans-loading terminals, in-transit rail, barge, and truck shipments, and coal-fired power plants is that spontaneous combustion can be predicted and prevented if conditions are changed (physically and chemically) to prevent the thermal runaway.
Are all coal types and coal ranks susceptible to spontaneous combustion?
Yes, all types of coal are vulnerable to spontaneous combustion, however it is the lower quality coals such as peat, brown coal, lignite, and sub-bituminous that are most prone. These coal types are known for having higher moisture contents, higher volatile contents, and lower carbon and BTU contents. Combine these attributes with the fact that these lower quality coals are far more friable (easily reduced to powder), and it is easy to understand why they are far more prone to spontaneous combustion. The higher moisture content causes an exothermic response when the moisture dissipates into the dryer air. The higher volatile content provides an inherent instability to the substrate. Their highly friable nature not only increases the available surface area for oxidation, it also provides excellent insulating properties that prevent heat from dissipating during oxidation. All of these factors together create a perfect environment for combustion to occur spontaneously.
Sub-bituminous coal mined from the Powder River Basin of Wyoming and Montana is notorious for its susceptibility to spontaneously combust. Because PRB coal is low in sulfur and extremely economical, it is in high demand all over the world. As more and more PRB coal is mined from this region, trans-loading operations and coal-fired power plants who were used to handling bituminous coal have had to change the way they handle and store coal in order to reduce spontaneous coal combustion risks.
Many users who are familiar with PRB coal know it’s not a matter of “if,” but “when” you will have a spontaneous coal fire. It is common to see PRB coal smolder and catch fire in storage piles and in bunkers or silos at mines, terminals, and power plants. There are even times sub-bituminous coal, regardless of origin, has been delivered to a power plant with the rail car or barge partially on fire.

Spontaneous coal fires during transport
What is MinTech doing to help manage spontaneous coal combustion?
Over the last two decades, MinTech has invested enormous resources in the study of PRB coal and other low grade coals in an effort to understand the mechanism of action. From this research, MinTech has developed a comprehensive line of inhibitors designed to prevent spontaneous coal combustion. By understanding the mechanism that drives spontaneous heating and thermal runaway, MinTech has been able to incorporate these inhibitors into products and programs that prevent spontaneous combustion of coal during the mining, transport, and storage processes.
MinTech inhibitors are able to slow the oxidative process of coal both physically and chemically, depending on the application and conditions.
Inhibition via physical control methods includes the incorporation of our chemistries into our MinCryl™ and MinTopper™ lines of coal pile sealants and bunker sealants. The inhibitors in these products protect coal storage from moisture and oxygen exchange, reducing heat formation. A secondary benefit of this slowdown to the auto-oxidation process is the conservation of heat value (BTU content). By conserving the heat value of the coal during short and long-term storage, coal-fired power plants are able to generate more electricity with less coal. Even small improvements in efficiency at this level can result in millions of dollars saved each year.

Application of pile sealant to prevent spontaneous combustion during storage
Inhibition via chemical control methods includes the incorporation of our chemistries into our CoalTrol™ and DustNot™ lines of process dust suppressants. Chemical inhibitors like MinTech’s FlameFreeze™ anti-oxidant technology, directly interrupt the oxidation and combustion processes. MinTech is the only company capable of combining our spontaneous combustion inhibitors into process dust suppression solutions, allowing our customers to control dust while simultaneously reducing the risk of spontaneous combustion throughout their coal handling processes. Our proven programs include full-body inhibitor treatments for rail loading, barge loading, and stack out operations.

Coal reclaimed from the coal-yard treated with spontaneous combustion inhibitors
Can existing dust suppression systems be utilized to apply MinTech spontaneous combustion inhibitors?
Yes, MinTech can easily retro-fit any dust suppression system to allow it to apply MinTech CoalTrol and DustNot products. The process is simple, starting with a free site survey performed by one of MinTech’s engineers. During the upgrading process, MinTech engineers will work with MinTech chemists to select the best dust suppressant/spontaneous combustion inhibitor blend for your exact needs. The equipment upgrade will meet your needs and your budget. The chemicals employed will be compatible with your water, formulated for your coal, and developed to meet your safety and environmental health requirements.
Who will service my dust suppression system?
MinTech services every dust suppression system we install at no charge. We know that dust suppression and spontaneous combustion chemicals will only perform properly if they are applied properly. That’s why our service technicians perform routine maintenance on every system we service.
For more information on our services or to schedule a free on-site demonstration, please contact us.
